A guide to federal accessibility requirements for medical diagnostic equipment — and why the compliance window is closing in 2026.
2026 Compliance Deadlines Are Here. As of mid-2026, federal compliance deadlines for accessible medical diagnostic equipment are in effect. Healthcare facilities that have not yet evaluated their equipment against the Access Board’s MDE Standards should do so now.
The Regulatory Foundation
The Americans with Disabilities Act of 1990 (ADA) and Section 504 of the Rehabilitation Act of 1973 are federal civil rights laws that prohibit discrimination against individuals with disabilities in everyday activities, including equal access to medical services.
While the ADA passed in 1990, concrete specifications for medical devices intended to remove barriers for patients with mobility disabilities did not emerge until the Affordable Care Act of 2010, which directed the U.S. Access Board to develop those standards. The Access Board began its medical device rulemaking in 2012, and in January 2017, those standards were published as 36 CFR Part 1195 in the Federal Register.
Notably, the U.S. Department of Veterans Affairs adopted the Access Board regulations as its procurement standard early on, and several states have also indicated they intend to adopt these new standards to regulate access to healthcare by patients with mobility disabilities. As of 2024, both the U.S. Department of Health and Human Services (HHS) and the Department of Justice (DOJ) have issued final rules making these standards enforceable federal law. See the section below.
What Changed: Federal Enforcement Is Now Reality
In 2024, both HHS and the DOJ finalized rules making the Access Board MDE Standards enforceable. These are not proposed rules. They are final and in effect.
HHS Section 504 Final Rule — effective July 8, 2024
On May 1, 2024, HHS finalized a rule that prohibits discrimination on the basis of disability, clarifying and strengthening civil rights protections for people with disabilities, addressing discrimination in medical treatment, and adding enforceable standards for accessible medical diagnostic equipment. The rule applies to all public and private entities that receive any form of HHS federal funding, including doctors, dentists, hospitals, clinics, emergency rooms, and any provider participating in Medicare or Medicaid.
DOJ Title II Final Rule — published August 9, 2024
On August 9, 2024, the DOJ issued a final rule updating the regulation for Title II of the ADA to improve access to medical diagnostic equipment for people with disabilities. The rule clarifies how public entities that use MDE — such as hospitals and health care clinics operated by state or local governments — can meet their obligations to ensure accessibility under the ADA. The rule adopts the U.S. Access Board’s MDE Standards as the technical standard for state and local government entities.
The two rules are intentionally parallel. Because Title II of the ADA is modeled on Section 504 and courts have interpreted them to be consistent, the Title II rule imposes virtually the same obligations on public entities as HHS’s rule imposes under Section 504. Entities that comply with the Section 504 rule as to MDE will necessarily comply with the Title II rule as well.
Enforcement Deadlines: A 2026 Action Timeline
Compliance is phased, with two critical deadlines arriving in mid-2026:
| Requirement | Who It Applies To | Date to Be Compliant By |
|---|---|---|
| All newly acquired MDE must meet Access Board standards | All HHS-funded recipients (Section 504) | July 8, 2024 (past) |
| All newly acquired MDE must meet Access Board standards | State & local government entities (DOJ/Title II) | Oct. 8, 2024 (past) |
| At least one accessible exam table and one accessible weight scale required | State & local government entities (DOJ/Title II) | July 8, 2026 |
| At least one accessible exam table and one accessible weight scale required | State & local government entities | Aug. 9, 2026 |
Sources: ADA.gov Final Rule Fact Sheet, Aug. 2024; HHS.gov Section 504 Fact Sheet, May 2024
Beyond the exam table and weight scale minimums, general scoping requirements apply for most healthcare providers: at least 10% — or a minimum of one unit — of each type of MDE in use must be accessible. For practices specializing in conditions affecting mobility, such as rehabilitation or physical therapy, this increases to 20% of each type of MDE.
The new rule also requires covered healthcare practices to adequately train staff to operate accessible MDE and to assist with the transferring and positioning of individuals with disabilities. This may require the purchase of transfer boards, Hoyer lifts, or other assistive equipment needed for the safe transfer of individuals.
Who Is Covered
One of the most significant aspects of the 2024 rules is how broadly they apply.
As of July 8, 2024, HHS has in place new requirements for the accessibility of MDE used by public and private entities that receive funding from HHS. The requirements apply to doctors, dentists, hospitals, clinics, emergency rooms, and other health care providers that use medical diagnostic equipment in providing their services. Entities that participate in Medicare, Medicaid, or other grant or funding programs from HHS are covered.
The DOJ Title II rule currently applies specifically to state and local government-operated healthcare facilities. However, because the DOJ is responsible for regulation under Title III of the ADA and, in conjunction with HHS, for Rehabilitation Act regulation, these rules will likely be made applicable to all recipients of federal financial assistance and eventually privately owned public accommodations. Private practices should not wait for that expansion to begin preparing.
Private Practice Note: If your practice accepts Medicare or Medicaid — and almost all do — the HHS Section 504 rule already applies to you. The question is not whether these requirements are coming. It’s whether your equipment is ready.
The Technical Standards: What Accessible MDE Looks Like
The Access Board divided MDE into categories based on how patients use the equipment. These categories have not changed — but they are now enforceable federal standards, not merely recommended guidelines.
The Access Board has divided the Standards for Accessible Medical Diagnostic Equipment into separate technical criteria based on how the equipment is used by the patient:
- M301: Equipment used in supine, prone, or side-lying positions — general exam tables, GYN/OB equipment
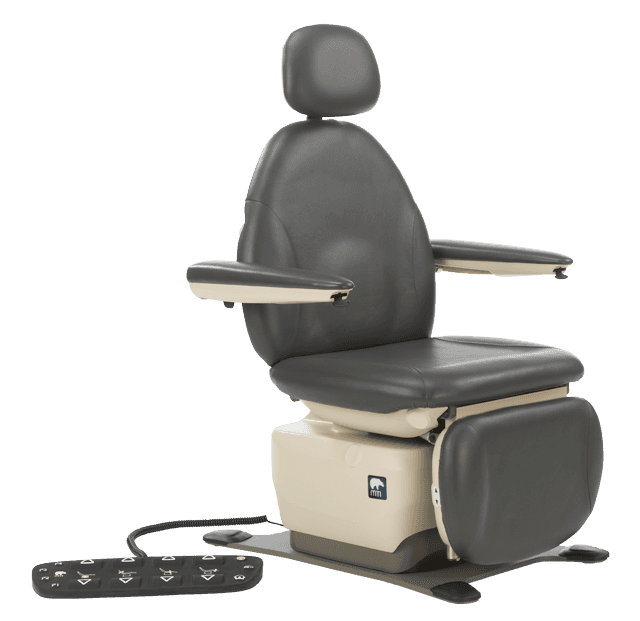
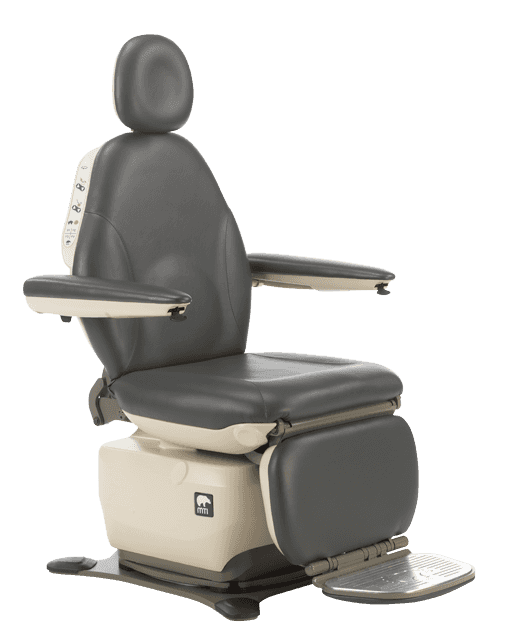
- M302: Equipment initially accessed from a seated position — ENT, podiatry, dermatology, ophthalmology, oral surgery chairs
- M303: Equipment accessed while the patient remains in a wheeled mobility device — mammography equipment, weight scales
Key Technical Specifications
| Spec | Requirement | Applies To |
|---|---|---|
| Transfer height (low) | 17″–19″ from floor, measured uncompressed | M301 & M302 |
| Transfer height (high) | At least 25″ | M301 & M302 |
| Transfer surface size | Minimum 21″ wide × 17″ deep | M302 only |
| Transfer sides | 2 adjoining sides (front & side); exception for fixed-footrest chairs | M302 |
| Transfer support | Min. 15″ length, max 1.5″ from surface, covers min. 13.5″ depth | M305.1–5 |
The Case for Compliance
Accessible medical equipment expands the range of patients a facility can serve effectively. For individuals with mobility disabilities, equipment that accommodates their needs means fewer barriers to care, more complete examinations, and better health outcomes overall. That is good for patients and good for practice.
It is also worth noting that while HHS and the DOJ have not outlined a specific enforcement action process for non-compliance, facilities that do not meet the MDE Standards may open themselves to litigation from individuals who experience barriers to care. Ensuring your equipment meets the applicable standards is a straightforward way to reduce that exposure.
MTI has 11 procedure and exam chair models that meet all applicable requirements under 36 CFR Part 1195 Standards for Accessible Medical Diagnostic Equipment for standard, swivel, and mobile bases, including the 829, 830, 550, and others across the MTI product line.
The U.S. Access Board’s Updated Low Transfer Height Standard
It is worth noting that the U.S. Access Board has published an updated standard specifying a low transfer height of 17″ — a lower threshold than the 17″–19″ range currently enforced by HHS and the DOJ. This updated standard reflects the Access Board’s ongoing work to improve accessibility specifications, but it has not yet been adopted into the enforceable rules issued by either agency. The DOJ has indicated it is considering a supplemental rulemaking to address this in the future.
MTI’s new 830-17 is designed to meet the Access Board’s 17″ specification. For facilities that want to get ahead of where the standards may be heading, or that simply want to provide the most accessible experience possible for patients with mobility disabilities, the 830-17 offers that option — while still meeting all current HHS and DOJ enforceable requirements.
What Healthcare Facilities Should Do Now
With the July and August 2026 deadlines arriving imminently, facilities should be moving on several fronts simultaneously:
- Audit your current MDE inventory against Access Board standards
- Identify which equipment categories apply to your practice (M301, M302, or M303)
- Prioritize acquiring at least one accessible exam table and one accessible weight scale if not already in place
- Confirm your scoping obligations (10% for general practices, 20% for mobility-focused practices)
- Verify that accessible equipment is placed on an accessible route and maintained in working order
- Train staff on accessible MDE operation and safe patient transfer assistance
- Review vendor contracts to ensure future acquisitions continue to meet MDE Standards